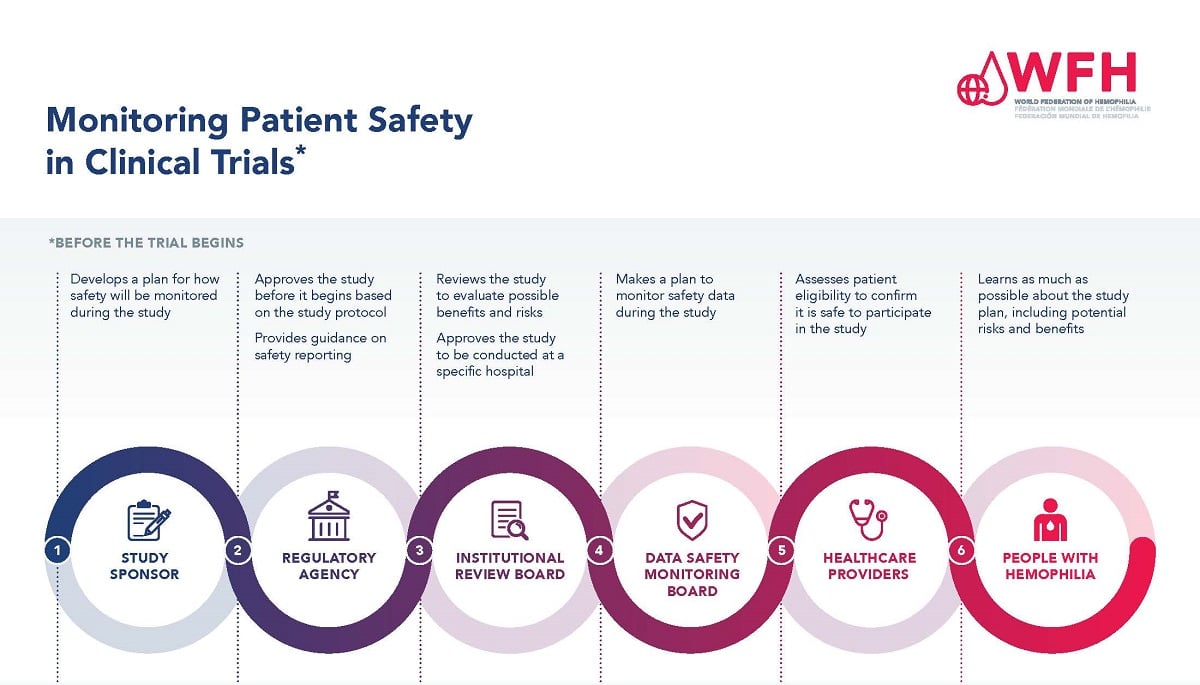
Safety Population Clinical Trial . clinical trials are an important source of safety data that contribute to the totality of safety information available to. the safety population is used for the analysis of safety, including adverse events, toxicity and laboratory.
from elearning.wfh.org
clinical evaluation of diagnostic agents; Frequent and thorough monitoring of patient safety is a requirement of clinical trials research.the efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the.
Monitoring Patient Safety in Clinical Trials eLearning Platform
Safety Population Clinical Trial safety is an essential part of the evaluation of new medications and competing risks that occur in most clinical.clinical evaluation of diagnostic agents; The extent of population exposure to assess clinical. the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the.
From elearning.wfh.org
Monitoring Patient Safety in Clinical Trials eLearning Platform Safety Population Clinical Trialthe efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. the safety population is used for the analysis of safety, including adverse events, toxicity and laboratory. clinical trials are an important source of safety data that contribute to the totality of safety information available to. Frequent and thorough monitoring of patient. Safety Population Clinical Trial.
From www.semanticscholar.org
Figure 3 from Approaches to Phase 1 Clinical Trial Design Focused on Safety Population Clinical Trial The extent of population exposure to assess clinical. Frequent and thorough monitoring of patient safety is a requirement of clinical trials research.clinical evaluation of diagnostic agents; safety is an essential part of the evaluation of new medications and competing risks that occur in most clinical. the psap outlined proactive plans for routine aggregated analyses of clinical. Safety Population Clinical Trial.
From www.researchgate.net
Patient disposition. *Safety population comprises all patients who Safety Population Clinical Trial the safety population is used for the analysis of safety, including adverse events, toxicity and laboratory.clinical evaluation of diagnostic agents; Frequent and thorough monitoring of patient safety is a requirement of clinical trials research. safety is an essential part of the evaluation of new medications and competing risks that occur in most clinical. the aims. Safety Population Clinical Trial.
From www.lymphoma.org.au
Understanding Clinical Trials Lymphoma Australia Safety Population Clinical Trial the aims of this article are (1) to describe a general framework for repeated monitoring of safety events in clinical.the efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the.clinical evaluation of. Safety Population Clinical Trial.
From www.researchgate.net
Trial participant recruitment and randomization. The safety population Safety Population Clinical Trial the safety population is used for the analysis of safety, including adverse events, toxicity and laboratory.the efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. the aims of this article are (1) to describe a general framework for repeated monitoring of safety events in clinical. clinical trials are an. Safety Population Clinical Trial.
From crfweb.com
The Clincial Trial Process Stepbystep approach Safety Population Clinical Trialthe efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. the aims of this article are (1) to describe a general framework for repeated monitoring of safety events in clinical. The extent of population exposure to assess clinical.clinical evaluation of diagnostic agents; clinical trials are an important source of. Safety Population Clinical Trial.
From www.researchgate.net
Summary of safety (safety population) Download Table Safety Population Clinical Trial The extent of population exposure to assess clinical.the efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the.clinical evaluation of diagnostic agents; Frequent and thorough monitoring of patient safety is a requirement of clinical trials research. the safety population is used for the analysis of safety, including adverse events, toxicity. Safety Population Clinical Trial.
From www.youtube.com
Patient Safety in Clinical Trials YouTube Safety Population Clinical Trialthe efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. safety is an essential part of the evaluation of new medications and competing risks that occur in most clinical. Frequent and thorough monitoring of patient safety is a requirement of clinical trials research. the psap outlined proactive plans for routine aggregated. Safety Population Clinical Trial.
From www.slhn.org
Cancer Clinical Trials Cancer Care St. Luke's Cancer Center Safety Population Clinical Trial Frequent and thorough monitoring of patient safety is a requirement of clinical trials research.the efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. The extent of population exposure to assess clinical. the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the. the aims. Safety Population Clinical Trial.
From www.slideserve.com
PPT SAFETY MONITORING IN CLINICAL TRIALS PowerPoint Presentation Safety Population Clinical Trial clinical trials are an important source of safety data that contribute to the totality of safety information available to. the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the. The extent of population exposure to assess clinical. Frequent and thorough monitoring of patient safety is a requirement of clinical trials research.. Safety Population Clinical Trial.
From www.researchgate.net
Flow diagram of the clinical trial, showing the 3 populations analysed Safety Population Clinical Trial safety is an essential part of the evaluation of new medications and competing risks that occur in most clinical. the aims of this article are (1) to describe a general framework for repeated monitoring of safety events in clinical. the safety population is used for the analysis of safety, including adverse events, toxicity and laboratory. the. Safety Population Clinical Trial.
From www.quantics.co.uk
What is a Safety Population in a Clinical Trial? Quantics Biostatistics Safety Population Clinical Trial the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the. The extent of population exposure to assess clinical. safety is an essential part of the evaluation of new medications and competing risks that occur in most clinical. the aims of this article are (1) to describe a general framework for. Safety Population Clinical Trial.
From powerfulpatients.org
Diversity in Clinical Trials Benefits Everyone Patient Empowerment Safety Population Clinical Trial the safety population is used for the analysis of safety, including adverse events, toxicity and laboratory. clinical trials are an important source of safety data that contribute to the totality of safety information available to. The extent of population exposure to assess clinical.the efficacy and safety of medicinal products should be demonstrated by clinical trials which. Safety Population Clinical Trial.
From www.slideserve.com
PPT Safety Part I Clinical Trial Safety and Safety Safety Population Clinical Trial the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the.clinical evaluation of diagnostic agents; clinical trials are an important source of safety data that contribute to the totality of safety information available to. Frequent and thorough monitoring of patient safety is a requirement of clinical trials research. the. Safety Population Clinical Trial.
From www.researchgate.net
Adverse Events during Treatment Safety Populations in Trials Safety Population Clinical Trial the aims of this article are (1) to describe a general framework for repeated monitoring of safety events in clinical. the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the.the efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. safety is an. Safety Population Clinical Trial.
From www.massbio.org
Balancing the Scale How Bristol Myers Squibb is Ensuring Clinical Safety Population Clinical Trialclinical evaluation of diagnostic agents; The extent of population exposure to assess clinical. the aims of this article are (1) to describe a general framework for repeated monitoring of safety events in clinical. the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the. Frequent and thorough monitoring of patient safety. Safety Population Clinical Trial.
From clinicaltrialsalliance.org.au
Major Initiatives ACTA Australian Clinical Trials Alliance Safety Population Clinical Trial the psap outlined proactive plans for routine aggregated analyses of clinical trial safety data to define the. the safety population is used for the analysis of safety, including adverse events, toxicity and laboratory. Frequent and thorough monitoring of patient safety is a requirement of clinical trials research.the efficacy and safety of medicinal products should be demonstrated. Safety Population Clinical Trial.
From pharmaguidances.com
Phases of clinical trials Pharmaceutical Guidance Safety Population Clinical Trialthe efficacy and safety of medicinal products should be demonstrated by clinical trials which follow the. clinical trials are an important source of safety data that contribute to the totality of safety information available to. Frequent and thorough monitoring of patient safety is a requirement of clinical trials research.clinical evaluation of diagnostic agents; safety is. Safety Population Clinical Trial.